Anesthesia
and its monitoring in rabbits
Esther van Praag, Ph.D.
MediRabbit.com is
funded solely by the generosity of donors.
Every
donation, no matter what the size, is appreciated and will aid in the
continuing research of medical care and health of rabbits.
Thank you
|
Warning: this file
contains pictures that may be distressing for people.
|
Rabbits
are often considered as difficult animals in relation to general anesthesia.
This may relate to the fact that the doses needed to induce and maintain
anesthesia and those producing toxic effects are close to each other, and to
the variety of observed secondary effects related to stress and cardiac or
respiratory reactions. Anesthesia in rabbits is, nowadays, considered as a
safe procedure, when a minimum of safety measures is taken. This includes a
complete check-up of the rabbit, the correct anesthetics agents, no
malfunctioning equipment and doing the different steps on a calm rabbit. Since
rabbits are unable to vomit, there is no need to withhold the food and water
longer than 2 to 4 hours before the planned surgical procedure. In fact,
rabbits that do not eat over a longer period of time before the surgery show
an increased tendency of becoming hypoglycemic during surgery or get
post-surgical disturbances of the gastro-intestinal tract due to dysbiosis.
Growth of pathogenic bacteria leads to the development of enterotoxaemia. The
rate of recovery appears, furthermore, slowed down in rabbits whose food was
taken away hours prior to surgery. It is thus advised to keep food available
up to 2 to 4 hours (depending on the surgical procedure) before anesthetic preparations
are started. This way the oral cavity does not contain food rests and that
the stomach is not overloaded and distended. Food and water should be
available immediately after the rabbit recovers from the anesthesia. Pre-anesthetic steps are often useful in
the preparation of surgical anesthesia. Click
here for details. Induction of general anesthesiaThe choice of the anesthetic drug and its
way of administration depend on the health condition of the rabbit, and the
length of the surgery. Intravenous administration of anesthetic drugs is the
least recommended way since the toxic doses of some anesthetic agents is
close to that needed for surgical anesthesia.
When
the injected subcutaneous or intramuscular way is chosen, higher doses of
anesthetics must be used. As a consequence, waking-up time is slower and the
risk of hypothermia is increased if no heating pad is used, or fluids are not
administrated at body temperature. Some compounds also have known side
effects or adverse effects, e.g. rabbits regularly anesthetized with the
ketamine/xylazine mixture showed increased rates of heart disease and an
increased death rate. It is necessary
to take those adverse or side effects into consideration on an individual
basis, according to the health state of the rabbit. Some rabbits also
appeared to wake up more slowly and with more difficulty from gas anesthesia
after need of regular teeth trimming. A list
of anesthetic drugs safe for use in rabbits, including their side effects, is
available. Click
here. Gas anesthesia is commonly used in
rabbits too. If this method is chosen, pre-anesthesia is necessary. Indeed,
rabbits will often start to struggle and attempt to escape when they smell
the agent, and may get hurt (e.g. spinal cord injury). Commonly used inhalant
gas agents are isoflurane and sevoflurane.
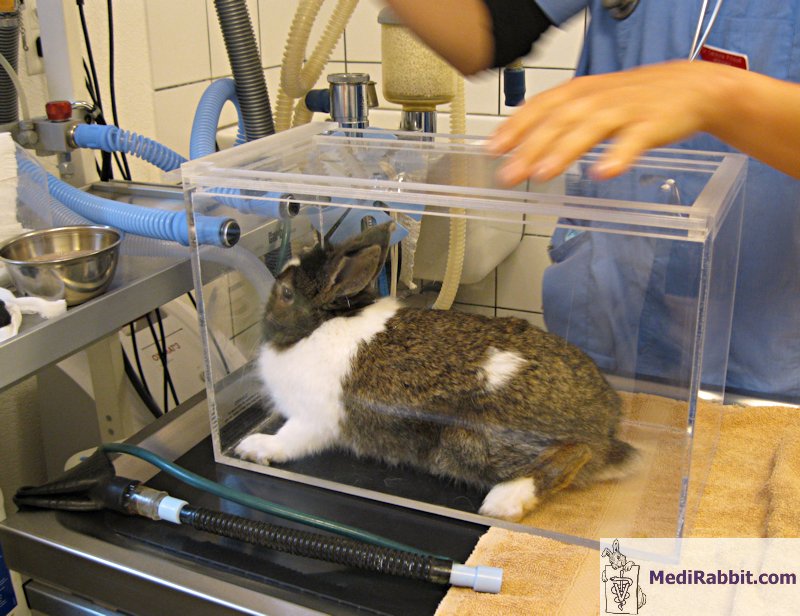
Gas anesthesia can be induced by placing
the rabbit in an induction chamber or by placing a mask tightly over its face
and holding the rabbit firmly.
Rabbits often try to retain their breath
independently of the method and the gas agent used. Rarely, bradycardia
(unusually slow heartbeat) or respiratory distress is observed.
Preparing the rabbit for surgery and monitoring vital
parameters during induction of anesthesia. Video taken by Debbie Hanson. When an induction chamber is used, it is
advisable to remove the rabbit once it shown signs of relaxation and place
him on a mask. If a nasal or face mask is used, it should be placed deep over
its face in order to minimize dead-space.
Induction of anesthesia in a rabbit placed in an induction
box. Video taken by Elena Grisafi Favre. The mask presents the advantage that it
can quickly be removed, and be replaced as soon as the animal starts
breathing normally again. In rare cases, when the rabbit refuses the breath
the anesthetic gas, the induction can be started with propofol or with
etomidate, before placing the mask, and continue the induction with the
chosen inhalant anesthetic.
Monitoring anesthesiaWhen
the induction phase is finished, preparations for surgery can be started.
This includes shaving, and disinfecting the skin. The depth of anesthesia
must be verified before placing a surgical drape over the animal. Reliable
methods in rabbits include: • Pedal
reflex, breath holding or hypoxia, and screaming: light depth; • Palpebral
(blink) reflex, ear pinch or left withdrawal reaction: medium depth; • Corneal
reflex: dangerously deep. When such depth is reached, cardiac arrest may
occur. Emergency measures should immediately be implemented, by reducing or
halting the administration of anesthetics. Exception: anesthesia with
medetomidine. The
level of surgical anesthesia is reached when reflexes to ear pinch and jaw
tone are lost. During
the anesthesia, the color of the mucous membranes (eye, lips tongue), the
respiratory rate, the heart beat and the rectal temperature should be
monitored. Monitoring vital parametersMonitoring of the respiratory rate, depth
and rhythm The
rate of respiration depends on the used anesthetic drug. The general tendency
is a decrease of the number of breaths per minute, to about 30 to 60. When
the rate is lower than 30 breaths per minute, or less that 50% of the normal
rate, there should be concern. Once the anesthesia and vital parameters are
stable, the respiration rate should remain regular and slow, though sudden
changes are not uncommon in rabbits Respiratory
rate can be monitored by observing the movement of the chest, when no
surgical drape is used. If this is not possible, respiration should be
monitored. It should be mentioned that some electric monitors for respiration
are unable to measure the rapid respiratory rate of rabbits and other small
animals, and thus become inefficient. The
physiological status of the rabbits can also be followed by monitoring the
exhaled CO2 (capnography). In this case, it is important to
note the general trend, and not individual numbers. If capnography is used,
it is important to take into account the dead-space of the equipment and mask
into the anesthetic equipment. Mainstream capnography is thus not advisable.
Side-stream capnography can be used, though the volume of the sampled gas may
be very large in comparison to the tidal volume. Monitoring
of the heart and heart rhythm It is
important to monitor the cardiac activity in anesthetized rabbits. Indeed,
pre-anesthetic examination and handling can cause a dangerous raise the level
of catecholamine. This can have disastrous consequences such as a lack of
correct oxygenation of the blood (hypoxemia) or an excess of CO2 in the blood (hypercapnia). Further problems seen in rabbits include
hypotension or an unusually slow heart rhythm (bradycardia).
The
heart beat rate of rabbits is rapid and may exceed 250 beats per minute. ECG
may thus be difficult, since the upper limits of the monitoring device are
reached (usually set at 250 or 300 beats per minute), except when
medetomidine or ketamine/medetomidine is used (decrease to 120-160
beats/min). Pulse
oximeters can be used to follow the heart rate and the level of oxygenation. The
values typically should remain higher than 90%; lower values typically
indicate that the level of oxygen in the blood is insufficient (hypoxemia). Reliable
signals are obtained when the probe is fixed to the tail, the ear, the
tongue, the hand or a digit of a limb in larger rabbits. Cardiac
arrest rarely occurs during anesthesia. This is an emergency situation that
leaves little time for intervention; in rabbits cardiac arrest is quickly
followed by respiratory arrest and death. Monitoring of the body temperature Control of body temperature of the rabbit
during anesthesia is essential due to: • The large surface area to body mass ratio;
leading to rapid loss of temperature; • Convective and radiant heat loss; • The effect of anesthetic agents on the
body temperature mechanisms; • Pre-surgical
and surgery itself. A good balance should be found between the minimum area
to shave at the surgery site, the minimum shaved area ensuring asepsis, and
the use of minimal quantities of disinfecting solutions.
Hypothermia tends to increase dangerously
the anesthetic depth during the surgical procedure. This may be accompanied
by hypoxia, acidosis, cardiac arrhythmia and a disturbance of the metabolism
of blood platelets. The body temperature of an anesthetized
rabbit can be monitored with an electronic thermometer, or a thermo-sensor
inserted deep in the rectum. The use of heating pads, hot water pads
or bottle, or convective heat sources help maintain body temperature during
the surgical procedure, during the waking-up and recovery phases. When fluid therapy
is necessary, the fluids must be heated at body temperature by warming them
in a microwave or passing the tube through a bowl of lukewarm water. AcknowledgementMany thanks to Amir Maurer, DVM
(Companion and Exotic Animal Veterinary Center, Holon, Israel), Elena Grisafi
Favre (la Colline aux Lapins,
Switzerland), and to Akira Yamanouchi (Veterinary Exotic Information Network,
Japan) for the permission to use their pictures to illustrate this page.
Thank you also to the veterinarians Thomas et Caroline Pilloud
(Cabinet vétérinaire du Brevil,
Boudevilliers, Suisse) for their cooperation. Further InformationDupras J, Vachon
P, Cuvelliez S, Blais D.
Anesthesia of the New Zealand rabbit using the the
combination of tiletamine-zolazepam and
ketamine-midazolam with or without xylazine. Can
Vet J 2001;42:455-60. Flecknell PA, John M, Mitchell M, Shurey C, Simpkin S. Neuroleptanalgesia in the rabbit. Lab Anim
1983;17:104-9. Flecknell P. BSAVA Manual of Rabbit
Medicine and Surgery, Gloucester, UK: British Small Animal Veterinary
Association 2000. Flecknell PA, Roughan
JV, Hedenqvist P. Induction of anaesthesia
with sevoflurane and isoflurane in the rabbit. Lab Anim
1999;33:41-6.
Harcourt-BrownOxford F. Textbook of Rabbit Medicine, UK:
Butterworth-Heinemann 2001 Hedenqvist P, Roughan
JV, Antunes L, Orr H, Flecknell
PA. Induction of anaesthesia with desflurane and isoflurane in the rabbit. Lab Anim 2001;35:172-9. Hillyer E.V. and Quesenberry
K.E. Ferrets, Rabbits, and Rodents: Clinical Medicine and Surgery, New York:
WB Saunders Co. 1997 Hobbs BA, Rolhall TG, Sprenkel TL,
Anthony KL Comparison of several combinations for anesthesia in rabbits. Am J
Vet Res 1991;52:669-74. Laber-Laird K. Handbook of Rabbit and
Rodent Medicine, Pergamon Veterinary Handbook Series)
Butterworth Heinemann 1996. Luo Y,
Russell GB, Griffith JW, Lang CM. Comparison of anesthesia induced by
ketamine-fentanyl combination and maintained by propofol
or etomidate in New Zealand white rabbits. Lab Anim
Sci 1995;45:269-75. Marini RP, Avison
DL, Corning BF, Lipman NS. Ketamine/xylazine/butorphanol: a new
anesthetic combination for rabbits. Lab Anim Sci 1992;42:57-62. Robertson SA,
Eberhart S. Efficacy of the intranasal route for
administration of anesthetic agents to adult rabbits. Lab Anim
Sci 1994;44:159-65. Scheller MS, Saidman
LJ, Partridge BL. MAC of sevoflurane in humans and the New Zealand white
rabbit. Can J Anaesth 1988;35:153-6.
Troitzsch D, Vogt S, Peukert
A. Study of long-term anesthesia in rabbits. Tierarztl
Prax 1996;24:519-21. Weinstein CH, Fujimoto JL, Wishner RE, Newton PO. Anesthesia of six-week-old New
Zealand White rabbits for thoracotomy. Contemp Top
Lab Anim Sci 2000;39:19-22. |
||||||||||||||||||||||
e-mail: info@medirabbit.com